|
4/19/2024 0 Comments What is absolute entropy 
When the entropy value is calculated for one mole of the substance in its standard state, the resulting absolute entropy is called the standard entropy. The superscript degree symbol (°) indicates that substances are in their standard states. Indeed, excellent results have been obtained for the free energy and the entropy. At any given temperature, the entropy value that is obtained in this way is called the substance’s absolute entropy or its third-law entropy. The term standard state is used to describe a reference state for substances, and is a help in thermodynamical calculations (as enthalpy, entropy and Gibbs free energy calculations). Given a discrete random variable, which takes values in the alphabet and is distributed according to : where denotes the sum over the variables possible values. This MC version of HS, called HSMC, is based on a relatively small number of efficiency parameters their number does not grow and their values are not changed as the number of the treated future atoms is increased (i.e., as the approximation improves) therefore, implementing HSMC for a relatively large number of future atoms (up to 40 in this study) is straightforward. In information theory, the entropy of a random variable is the average level of 'information', 'surprise', or 'uncertainty' inherent to the variables possible outcomes. The TP of target cell k is determined from the number of visits of future atoms to this cell during the simulation. A canonical metropolis Monte Carlo (MC) simulation is carried out over a portion of the still unvisited (future) volume thus providing an approximate representation of the N−N k as yet untreated (future) atoms. This means that standard entropy is really just the absolute entropy for a substance at 298 K. Recall that standard entropies are standardized at 298 K. 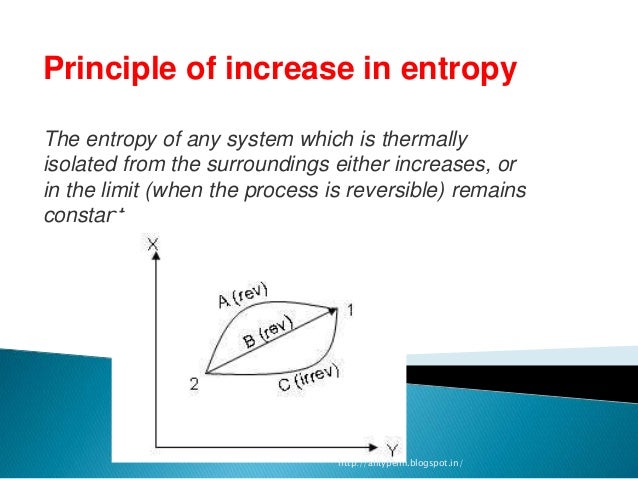
At step k, k−1 cells have already been treated, where among them N k are occupied by an atom. Absolute entropy is the total amount of entropy acquired when a pure substance is warmed from absolute zero to a specific temperature. At each step a transition probability (TP) is calculated and the product of all the TPs leads to P i. As in the preceding paper (Paper I), a probability P i approximating the Boltzmann probability of system configuration i, is calculated with a reconstruction procedure based on adding the atoms gradually to an initially empty volume, where they are placed in their positions at i in this process the volume is divided into cubic cells, which are visited layer-by-layer, line-by-line. A variant of the hypothetical scanning (HS) method for calculating the absolute entropy and free energy of fluids is developed, as applied to systems of Lennard-Jones atoms (liquid argon). According to the Boltzmann equation, entropy is a measure of the number of microstates available to a system. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
